
Editorially independent content, supported with advertising from Zeiss
June 2024
I. Paul Singh, MD; Joseph F. Panarelli, MD; David Eichenbaum, MD; and Mitchell A. Jackson, MD
In the Innovation Journal Club (IJC) series on Eyetube.net, host I. Paul Singh, MD, of The Eye Centers of Racine & Kenosha in Wisconsin, interviews leading experts from across eye care subspecialties about emerging innovations and technologies that may prove influential to the real-world practice of ophthalmology. The series is editorially independent (supported by advertising from multiple companies), which allows the discussions to be broad in scope and candid in presentation.
The following is a summary of three episodes in which Dr. Singh examined the relevance of real-world data on minimally invasive glaucoma surgery (MIGS) and minimally invasive bleb surgery (MIBS) with Joseph F. Panarelli, MD; spoke with retina specialist David Eichenbaum, MD, about gene therapy for the retina; and discussed laser scleral microporation with Mitchell A. Jackson, MD.
Despite their necessity, randomized, prospective studies are costly, time-consuming, and increasingly difficult to recruit patients for. In contrast, real-world evidence like retrospective analyses may not be the gold-standard, but if conducted well, they do provide valuable information about a treatment’s long-term safety and efficacy. Indeed, data from real-world studies “help to guide our decision-making process, but data doesn't dictate our decision-making,” said Joseph F. Panarelli, MD, in a recent episode of IJC.
Dr. Panarelli joined Dr. Singh in reviewing glaucoma patients’ outcomes following canaloplasty and trabeculotomy that were performed with the OMNI Surgical System (Sight Sciences, Inc.) post-cataract surgery by a single surgeon. Yadgarov et al assessed IOP outcomes and medication usage at 12 and 24 months in 171 eyes of 171 consecutive patients, with results stratified by mild, moderate, or severe open-angle glaucoma.1 The researchers also noted whether a secondary surgical intervention (SSI) was required.
In the study, there was a significant reduction in both IOP and medication usage from baseline after OMNI surgery at both 12 and 24 months (Figure 1). Perhaps not unexpectedly, patients with mild or moderate glaucoma achieved greater medication reduction and the ability to avoid SSI compared to the severe group (Figure 2). “In some patients,” Dr. Singh said, “[MIGS surgery] may not have enough power to get them off medications, especially the more advanced they are. What I thought was interesting about this study was that the medication burden reduction was greater in the mild-to-moderate patients.”
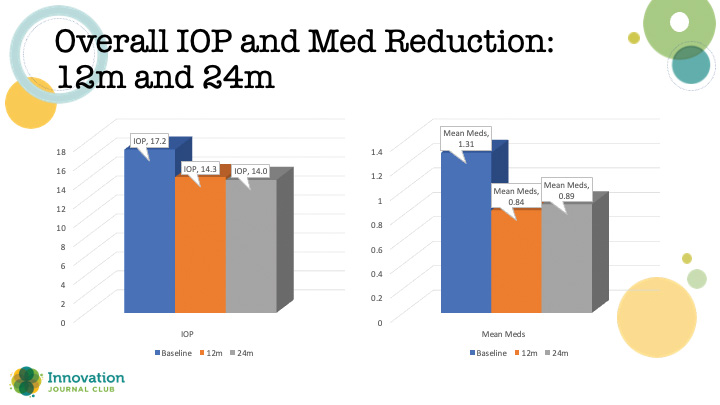
Figure 1. Reduction in IOP (left) and medication use (right) at 12 and 24 months after an OMNI procedure.
Dr. Panarelli also reviewed data from a systematic literature review of the Xen45 gel stent (Allergan, Inc./AbbVie) and its outcomes (postoperative changes in IOP and medication usage), and how outcomes correlated with patient demographics and other clinical factors.2 With an intentionally wide inclusion criteria to mimic an all-comers analysis, the researchers netted 111 studies. They found that the Xen45 implant was associated with a consistent reduction of IOP to the low-to-mid teens that remains stable as long as 36 months, and a frequent reduction in glaucoma medications. Furthermore, an analysis of IOP over time among studies reporting these data at interim time points demonstrated a predictable IOP-lowering effect in a pattern that mirrored the 12-month IOP data from Xen’s pivotal study in 2017 (Figure 3).3
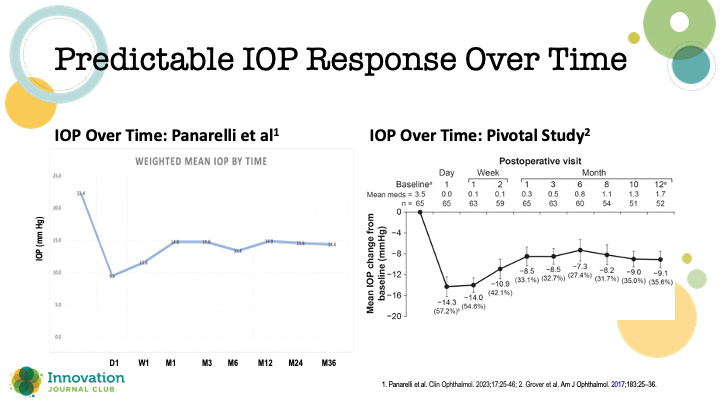
Figure 3. An analysis of IOP following Xen implantation at interim time points among studies in a systemic literature review (left) demonstrated a response curve similar to what was reported in the device’s pivotal study (right).
In addition to the predictability of its outcomes, Dr. Panarelli likes the reproducibility and low risk of the Xen procedure. “It’s one of those procedures that I feel good about. When I am being a little extra aggressive, I feel like I’m not taking on too much risk for a lot of these patients.”
1. Yadgarov A, Dentice K, Aljabi Q. Real-world outcomes of canaloplasty and trabeculotomy combined with cataract surgery in eyes with all stages of open-angle glaucoma. Clin Ophthalmol. 2023;17:2609-2617. eCollection 2023.
2. Panarelli JF, Vera V, Sheybani A, et al. Intraocular pressure and medication changes associated with xen gel stent: a systematic review of the literature. Clin Ophthalmol. 2023;17:25-46.
3. Grover DS, Flynn WJ, Bashford KP, et al. Performance and safety of a new ab interno gelatin stent in refractory glaucoma at 12 months. Am J Ophthalmol. 2017;183:25-36.
Although initially conceived to treat inherited retina disease, gene therapy is now being studied as a novel approach to manage common retinal pathologies like the dry and wet forms of age-related macular degeneration (AMD) and diabetic retinopathy (DR). In an episode dedicated to the state of the science around gene therapy in ophthalmology, Dr. Singh interviewed David Eichenbaum, MD, to learn about the different types of gene therapy, how they work, and why there is so much excitement for its potential.
According to Dr. Eichenbaum, the current research focus in gene therapy may be for common retinal diseases, but it has potential to impact all of ophthalmology’s subspecialties: “Gene therapy, even though it's focused on retina right now, has the potential to affect glaucoma, cornea—everything that we do, and it's in its early days.”
He explained that the form of gene therapy primarily being explored for retinal disease is gene augmentation (Figure 1). “In the case of most of our common retinal disease gene therapy programs, we’re directing the cell to produce a therapeutic protein, a ranibizumab-like product,” he told Dr. Singh. The therapeutic goal is to reduce (or eliminate) patients’ dependence on medications or injections, and thereby increase quality of life, to say nothing of boosting compliance and protecting the health of the ocular tissues (Figure 2).
Figure 2. Proponents hope that gene therapy can be a “one and done” option for addressing common retinal pathologies like AMD and DR.
But why has ophthalmology attracted so much attention for gene therapy research? Dr. Eichenbaum explained that the eye has several attributes that make it the perfect testing ground for gene therapy: It is small, easily accessible, is receptive to many forms of imaging technologies, can be examined directly and monitored for both therapeutic and adverse effects, and, most importantly, it self-contains any adverse reactions. As well, until recently, the only FDA-approved gene therapy across all of medicine was for RPE65-mediated retinal dystrophy (voretigene neparvovec; LUXTURNA, Spark Therapeutics). The latter demonstrated the viability of using viral vectors to deliver genetic material to retinal cells, thus serving as a starting point for investigations into the treatment of common retinal diseases.
Dr. Eichenbaum explained that gene therapy is essentially the use of a viral vector that contains genetic material that, when introduced into the nucleus of cells, reprograms their behavior (Figure 3). “In ophthalmology, we're using adenoviral-associated vectors because they have a low risk of genotoxicity, they can carry a large genetic payload, and they’re fairly well tolerated with efficient transduction,” he said. Importantly, there are ways to test if the changes to the DNA are having the desired effect on the disease.
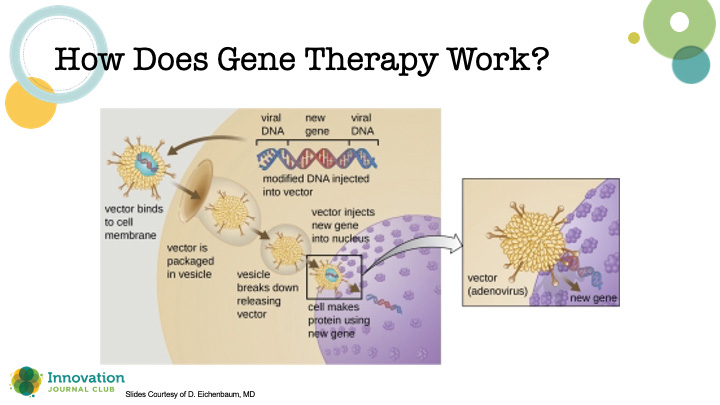
Figure 3. Schematic depicting the crucial steps for introducing genetic material to a host cell to induce a therapeutic effect.
There are three methods for delivering gene therapy currently under investigation. First is what Dr. Eichenbaum called the aspirational approach: intravitreal. This would be the preferred method of delivery since retina specialists are already well-versed in the technique. However, this method appears to have barriers, such as requiring a higher dose. As well, in early studies, there appears to be a higher rate of inflammation than other delivery methods (perhaps related to dosing), and researchers have noted physical barriers to delivering the product to the cells, like the inner limiting membrane.
Another method researchers are using is subretinal delivery, “because that seems to be the safest place to put it with regards to inflammation,” said Dr. Eichenbaum. One example of subretinal delivery is ABBV-RGX-314 (AbbVie/Allergan and Regenxbio), which is currently being investigated in a phase 3 study for wet AMD.
A third option for gene therapy delivery is suprachoroidal injection. This approach is in use in the phase 2 trial of ABBV-RGX-314 for treating DR, and Dr. Eichenbaum said that it may have potential down the line for treating macular degeneration. Its drawbacks, he said, are that the technique is “a little trickier and a little less routine than intravitreal injections,” and that the suprachoroidal injections are showing more inflammation in the phase 2 trial compared with subretinal injections. Yet, he feels that these are surmountable challenges.
Per Dr. Eichenbaum, most of the current research into ophthalmic gene therapies is focused on common retinal diseases that require injections, such as wet AMD, geographic atrophy, and DR, and for the most severe cases thereof—the patients who will benefit the most. The clinical trials of these products will answer the necessary questions of efficacy and safety, length and duration of effect, and whether they eliminate or just reduce the burden of injections. As part of these studies, researchers will also examine the best ways to mitigate the inflammatory response of gene therapy. He pointed out that as the ophthalmic population continues to age, these diseases will become more common, and thus the potential demand for gene therapy could be great.
Effectively addressing presbyopia may require a rethink of the mechanisms leading to loss of accommodative ability.
According to Mitchell A. Jackson, MD, a growing body of evidence suggests that models of accommodative ability focusing solely on movement of the ciliary body may be incomplete. Indeed, stiffening of the sclera in an aging eye and other extralenticular processes appear to be a major component in why patients lose the ability to focus at near as they age.
“The problem is there's cross-linking in the sclera, and it stops moving,” Dr. Jackson told Dr. Singh during a recent episode of IJC.
A novel treatment approach, known as laser scleral microporation (LSM), seeks to address presbyopia at its fundamental root cause. LSM describes a procedure in which an Er:YAG laser (VisioLite, Ace Vision Group) is used to create a diamond-shaped matrix of transconjunctival scleral micropores in four oblique quadrants (Figure 1). The procedure is performed bilaterally, and the goal is to un-crosslink the sclera.
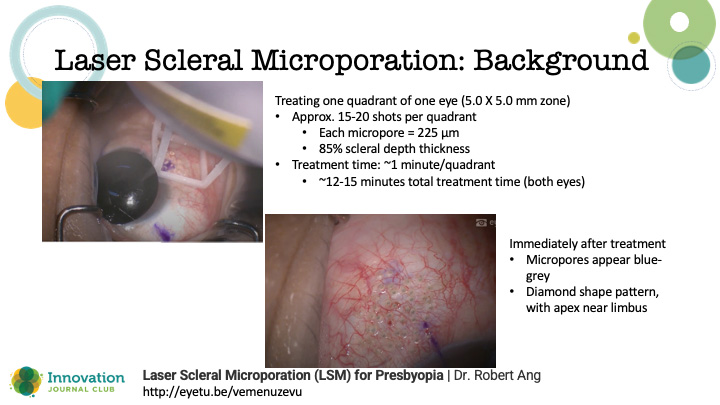
Figure 1. The LSM procedure is performed bilaterally, with the goal of un-crosslinking the sclera to allow a more complete dynamic range of focus across distance, intermediate, and near.
“By weakening the sclera, we get everything moving again, so we actually restore our dynamic range of focus versus our true accommodation,” Dr. Jackson said.
A recent study evaluated the dynamic range of focus at 24 months after bilateral treatments of LSM in 50 patients (average age = 52.6 years).1 The primary outcome was visual performance as assessed with ETDRS logMAR charts at distance, 60 cm, and 40 cm after 24 months.
In the study, no patient lost distance vision, intermediate vision was unchanged, and binocular distance-corrected near visual acuity had improved significantly by 24 months compared to baseline (Figure 2). “It's pretty amazing. It stays effective for at least 2 years, and it's at a level that matches anything we do with a premium lens and/or presbyopia eye drop that's been approved in the United States,” Dr. Jackson said.
As for the safety of the procedure with the current-generation laser (VisioLite, Ace Vision Group), moderate conjunctival vascularization and conjunctival edema may be evident on day 1 postoperatively, but these generally subside by week 1 (Figure 3). “This is really cosmetically acceptable. Eyes are white. Most patients are reading 20/40 or better uncorrected for near vision at day 1,” Dr. Jackson said.
Inclusion criteria for the study may offer some clues for patient selection, as patients were required to be using a reading add of at least 150+.
“You want people who are already in reading adds full-time and they're miserable,” Dr. Jackson explained. “I always call it the ‘LASIK-to-lens procedure.’ Most of the patients who undergo LSM are refractively oriented: they’ve had LASIK or whatever procedure to fix their distance, or if they're just an emmetrope, naturally born that way—what are you going to do until they get a cataract? You don't want to rush to intraocular surgery too soon in some of these patients, it might be a higher risk. With LSM, we have the perfect presbyopic or age-related treatment from that LASIK-to-lens procedure.”
While data collection is ongoing with the study, Dr. Jackson pointed out that Ace Vision Group is currently conducting trials with a next-generation laser that is coupled with an OCT device for even greater accuracy in achieving the desired scleral depth. The company is aiming to pursue a 510(k) approval for use of the next-generation laser in the United States.
1. Ang et al. Dynamic Range Of Focus Recovery In Emmetropic Presbyopes After Laser Scleral Microporation: 24 Month Pilot Study Outcomes. Presented at: European Society of Cataract and Refractive Surgeons annual conference. September 8-12, 2023. Vienna, Austria.
